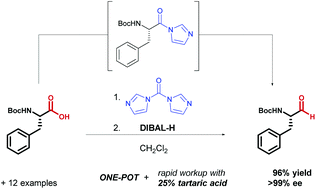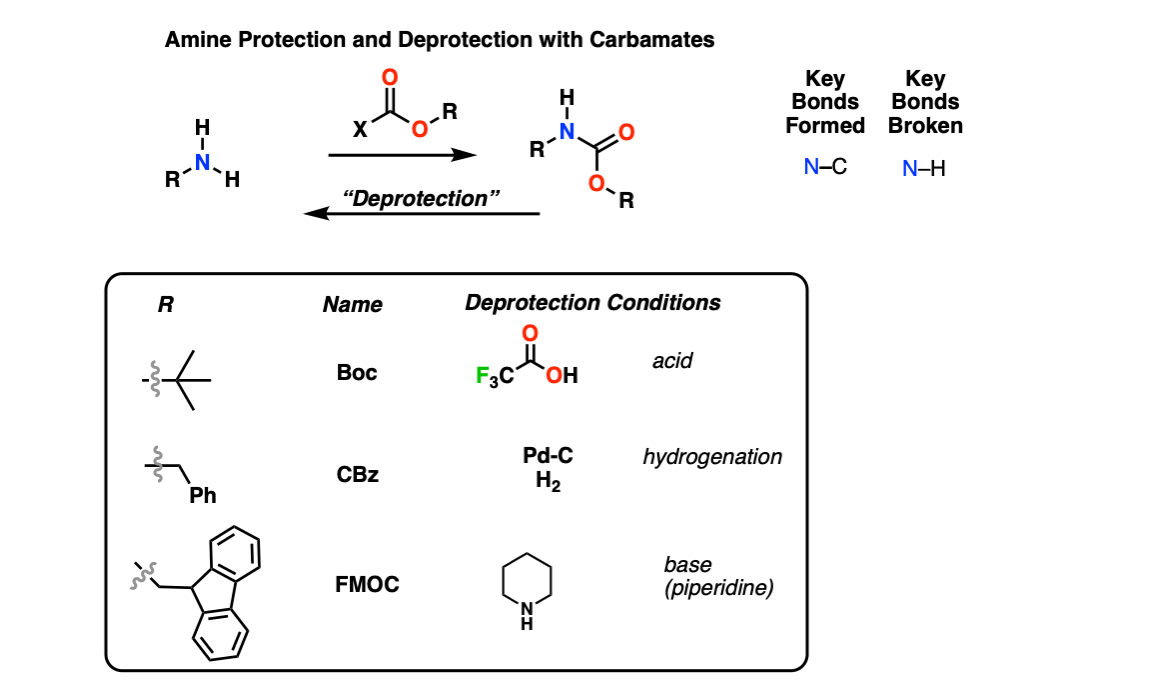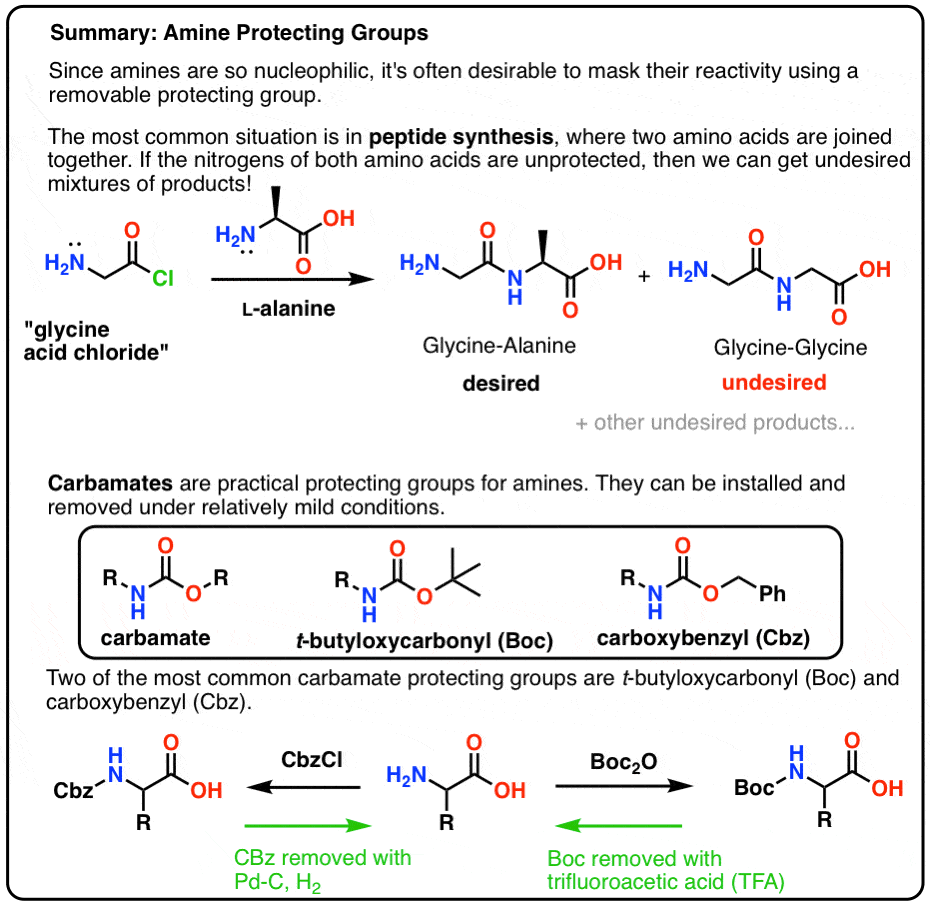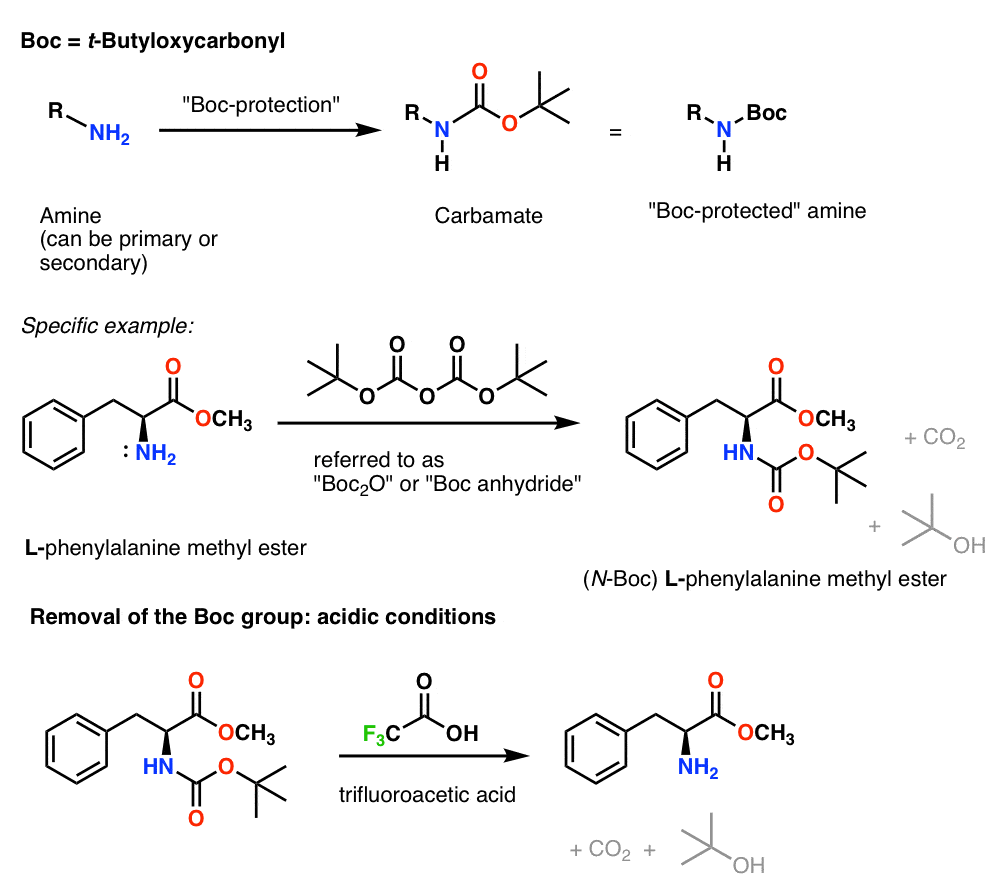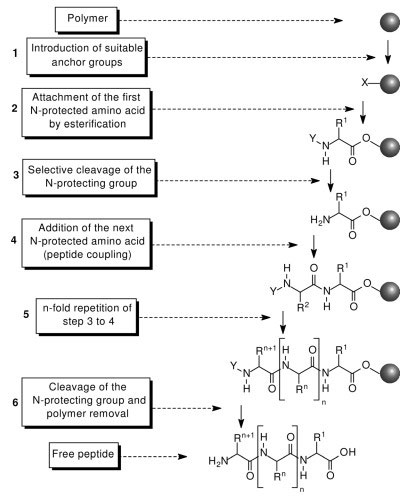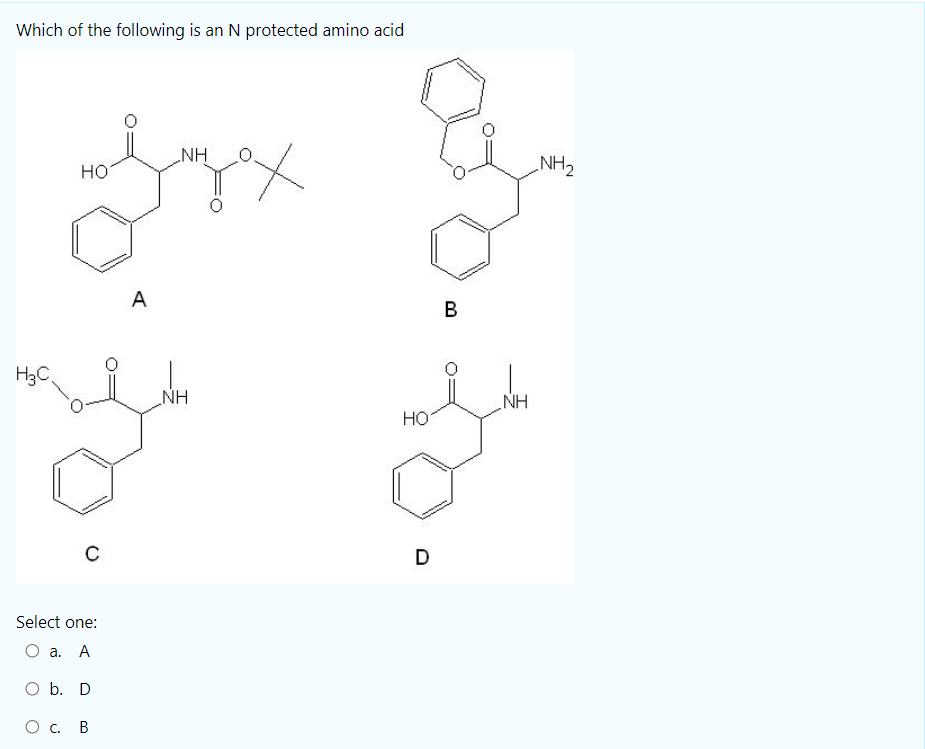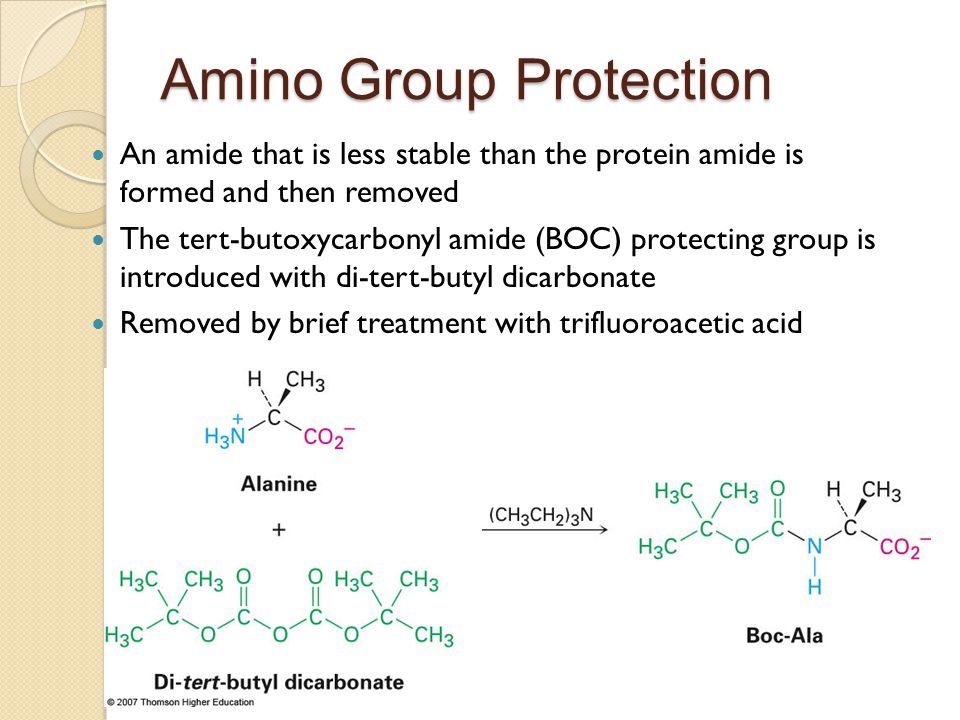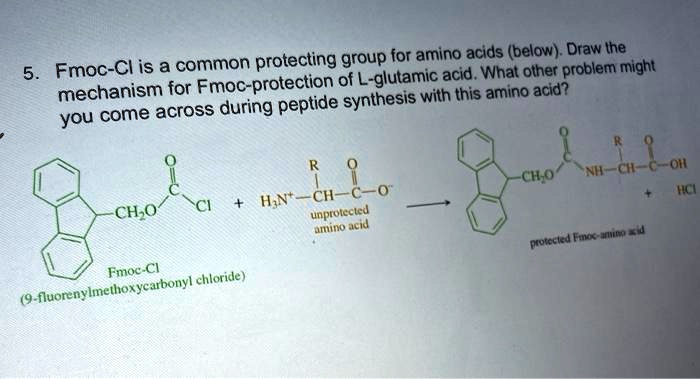
SOLVED: Fmoc-Cl is a common protecting group for amino acids. What other problem might arise with this amino acid during peptide synthesis?

An Efficient Synthesis of Nα-Protected Amino and Peptide Acid Aryl Amides via Iodine-Mediated Oxidative Acylation of Nα-Protected Amino and Peptide Thioacids

Catalytic Behavior of Mono‐N‐Protected Amino‐Acid Ligands in Ligand‐Accelerated C−H Activation by Palladium(II) - Salazar - 2020 - Angewandte Chemie International Edition - Wiley Online Library
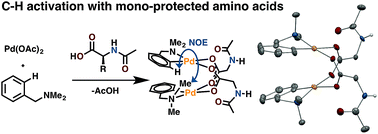
Mono-N-protected amino acid ligands stabilize dimeric palladium(ii) complexes of importance to C–H functionalization - Chemical Science (RSC Publishing)

Molecules | Free Full-Text | One-Pot and Catalyst-Free Transformation of N- Protected 1-Amino-1-Ethoxyalkylphosphonates into Bisphosphonic Analogs of Protein and Non-Protein α-Amino Acids

Direct amidations between N-Boc protected β-amino acid and C-protected... | Download Scientific Diagram

Protecting‐Group‐Free Amidation of Amino Acids using Lewis Acid Catalysts - Sabatini - 2018 - Chemistry – A European Journal - Wiley Online Library
L-Proline, 25 g, CAS No. 147-85-3 | Protected Amino Acids | Amino Acids and Amino Acid Derivatives | Organic & Bioorganic Chemicals | Chemicals | Carl Roth - International
L-Cysteine, 25 g, CAS No. 52-90-4 | Protected Amino Acids | Amino Acids and Amino Acid Derivatives | Organic & Bioorganic Chemicals | Chemicals | Carl Roth - International